Image by Tom Fisk from Pexels taken from Canva
Indonesia’s rapid growth places it among the fastest-developing nations within the Association of Southeast Asian Nations (ASEAN). With its burgeoning population and growing emphasis on healthcare, Indonesia is witnessing a remarkable expansion in its pharmacy retail sector.
Insight into the pharmacy chains landscape in Indonesia:
One of the driving forces behind the growth of Indonesia’s pharmacy retail sector is the increasing demand for over the counter (OTC) products. By 2040, the country’s OTC sales is forecasted to reach USD 6.31 bln with CAGR of 7.80%. The government’s implementation of the national health insurance JKN (Jaminan Kesehatan Nasional) has also played a pivotal role in shaping the industry landscape. With JKN increasing access to prescription and generic drugs, pharmacies proliferate around public and private hospitals, thereby augmenting market revenue. The rise of telemedicine companies such as Halodoc, Alodokter, SehatQ, and GoApotik, also supports the sales growth of medicines.
To meet the evolving needs of consumers, major pharmacy retail chains have entered the domain of e-commerce, offering their products through retail platforms and mobile applications. This move towards digitalization aligns with the growing trend of health-conscious individuals seeking nutritional products and supplements. The convenience of online shopping, combined with the increasing number of internet users in the country, has significantly boosted the online sales of healthcare products.
In response to changing consumer preferences, pharmacy retail chains are also focusing on providing value-added services such as personalized consultations, Rx prescriptions for minor health issues, and convenient home delivery services.
Indonesia counts more than 32,110 pharmacies within the country (according to Kemenkes – Indonesian MoH). West Java stands as the province with the highest number of pharmacies across Indonesia, totaling 5,114 stores. Following closely behind is East Java with 4,531 pharmacies, and Central Java, with 4,013 units. Top 5 pharmacy chains in the market account for > more than10% of total revenue, which include:
- Kimia Farma
- Apotek K-24
- AS Watsons Group
- Guardian Pharmacy
- Century Apotek Healthcare
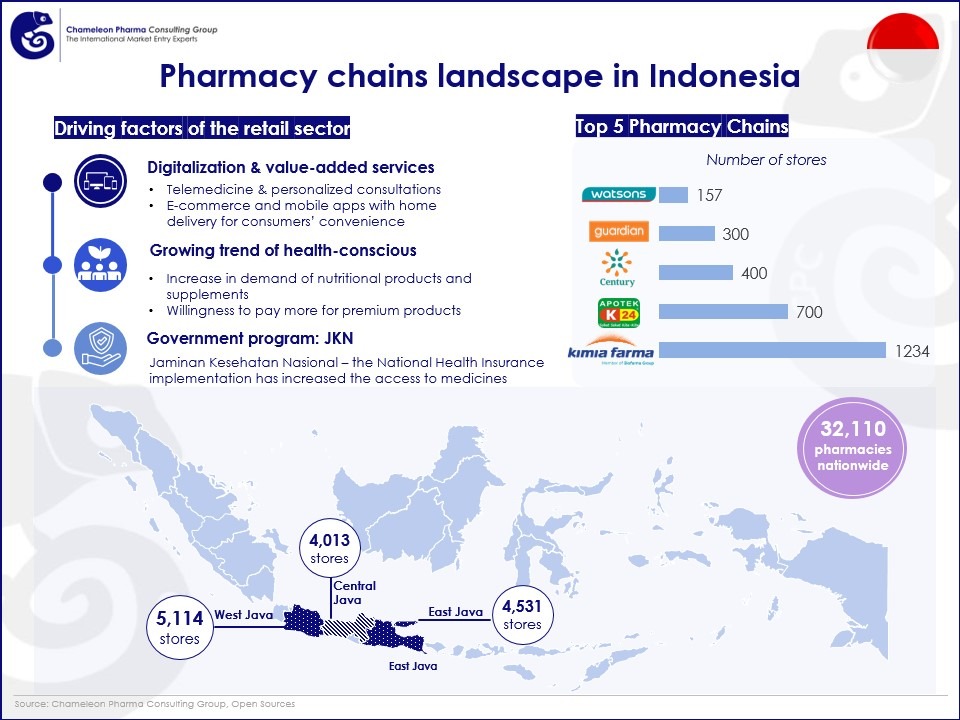
Figure 1. The pharmacy chains landscape in Indonesia
Distribution channels and margin structure in Indonesia:
Indonesia’s geographical complexity, characterized by its vast archipelago comprising over 17,000 islands and diverse terrain, poses substantial logistical hurdles for pharma distribution. Remote regions often suffer from inadequate road networks and deficient infrastructure, exacerbated by urban traffic congestion hampers the transportation and distribution of medical supplies.
Consequently, local small- and medium-sized pharma manufacturers typically opt to engage with specialized distribution (wholesale) partners to ensure seamless access to essential healthcare products nationwide. Meanwhile, larger corporations possess the resources and capabilities to independently distribute their own healthcare products.
The leading pharma distributors in Indonesia are listed as below:
- Merapi Utama Pharma:
- Anugrah Argon Medica (AAM)
- Anugerah Pharmindo Lestari (APL)
- Kalbe
- Kimia Farma
The distribution channels in Indonesia typically involve several layers, including manufacturers/importers, distributors, wholesalers, and retailers. Therefore, each mark-up might contribute to a high final retail price paid by the consumers. However, factors such as economies of scale, competition, and quality of service can also influence pricing dynamics in the Indonesian healthcare market.
Indonesia’s rapid growth and focus on healthcare are fueling an impressive expansion in its pharmacy retail sector within ASEAN. For international investors, this presents valuable opportunities, including dynamic distribution partnerships that can drive down costs and boost profit margins. With a deep understanding of Indonesia’s distribution landscape, investors can negotiate effectively with local distributors, streamlining the supply chain for greater efficiency and scalability. The future of pharmacy retail in Indonesia is bright, offering significant potential for those ready to seize the moment.
Chameleon Pharma Consulting Group specializes in internationalization, market entry and regulatory projects in the areas of consumer health, pharma, food supplements, medical devices, and cosmetics in the Middle East, as well as Latin America, Asia, the US, Europe, and CIS. We have completed over 300 international market entry projects, offering:
- Systematic Country and Product analysis (portfolio analysis),
- Systematic Local Partner Identification,
- Registration and Regulatory Affairs,
- Digital Transformation Services (strategy consulting, e-learning, social media analysis, SEO optimization and other).
For your individual questions please contact us by e-mail at service@chameleon-pharma.com.




