Photo by Waranont (Joe) on Unsplash
Thailand’s Consumer Health and Pharma market is expanding toward USD 14B by 2040, but growth is no longer uniform. Women’s health and pregnancy supplements are emerging as high-value niche shaping market entry strategy.
Rising Focus on Maternal Health in Thailand
Thailand’s Consumer Health and Pharma market is projected to reach approximately USD 14.1B by 2040, growing at around 8.3% CAGR, reflecting strong healthcare access and rising preventive care awareness.
A key recent development is the increasing national and regional focus on maternal nutrition, supported by international organizations and changing consumer behavior. Across Southeast Asia, governments and healthcare stakeholders are promoting early-stage supplementation (prenatal and pregnancy) to improve long-term health outcomes.
Women’s Health Supplements: A Fast-Growing OTC Segment
This shift is accelerating demand for Food Supplements targeting pregnancy and women’s health.
Key market dynamics include:
- Preventive care expansion: Women increasingly adopt supplements before and during pregnancy
- Medical recommendation influence: Doctors and pharmacists actively guide supplement use
- Premiumization trend: Imported EU brands benefit from perceived quality and safety
- Natural positioning: Strong demand for Phyto-based and clean-label products
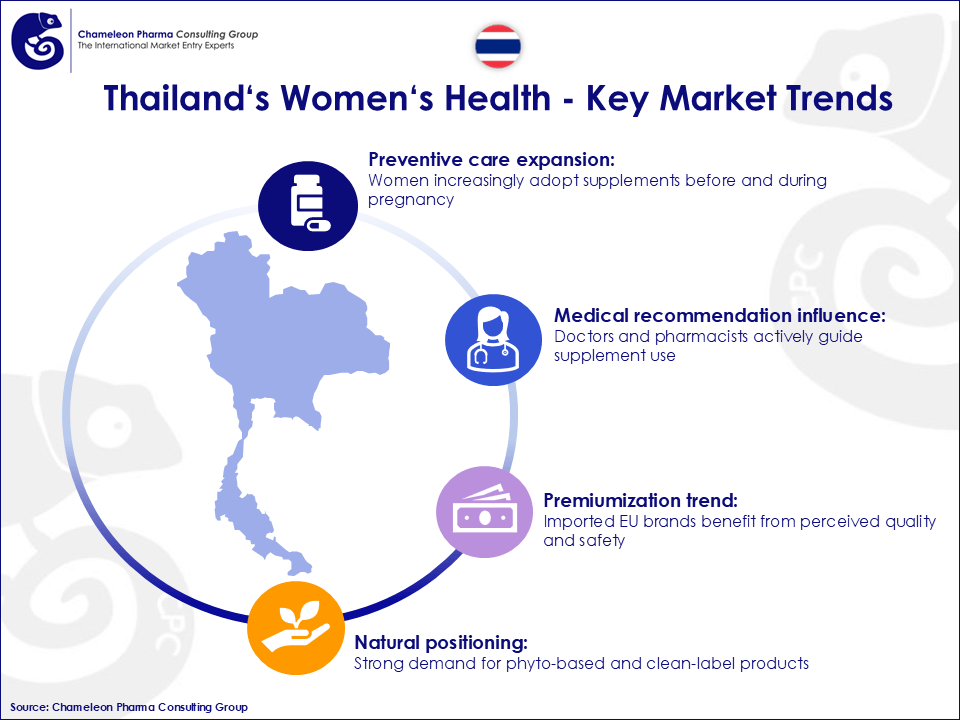
Figure 1: Thailand‘s Women‘s Health – Key Market Trends
Regulatory Implications for Market Entry
The Thai FDA plays a central role in enabling and controlling this growth, ensuring product safety and compliance. For Food Supplements, successful market entry depends on a clear understanding of key regulatory requirements, including:
- Correct classification (Food Supplement vs OTC)
- ASEAN-aligned dossier preparation
- GMP certification and quality standards
- Strict limitation on health claims
The regulatory framework in Thailand is relatively well-structured and increasingly harmonized with the regional framework. However, local expertise remains critical to avoid delays and ensure a smooth approval process.
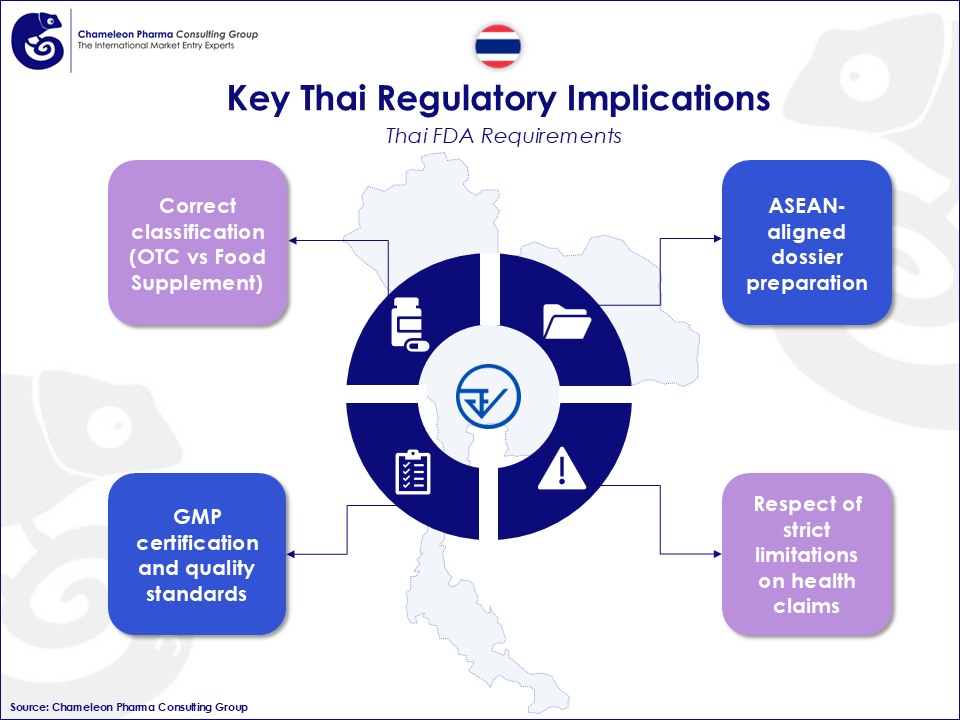
Figure 2: Key Thai Regulatory Implications
What This Means for Business Development Leaders
Women’s health and pregnancy Food Supplements are expected to outperform broader OTC segments in Thailand, supported by demographics, awareness, and healthcare policy alignment.
For Heads of Regulatory and Business Development, this trend signals a clear strategic window:
- Growing demand in the maternal health niche
- Strong alignment with preventive healthcare policies
- Opportunity to position premium, science-backed brands
Thailand can also serve as a gateway to ASEAN expansion, where similar trends are emerging.
To be best placed to capture this growth, companies need to combine regulatory readiness, local partnerships, and targeted positioning.
Chameleon Pharma Consulting Group (CPC) has over 20 years of experience in supporting Pharma, OTC, Medical Devices, Phyto, and Aesthetic Medicine companies. Having established own offices & local hubs across Latin America, Europe, Asia, the US/Canada, the Middle East, and the CEE/CIS regions is another advantage of CPC. With this local network and expertise gained from 300+ international projects and a team of 25 experts we offer our clients:
- Business Development, M&A, and Due Diligence
- Market Entry & Expansion: Systematic product and country analysis, market reports
- Strategic Partnering: Identifying local partners, acquisitions, or setting up own offices
- Regulatory & Registration: for drugs, MD, Derma, Aesthetic Medicine, etc.
- Market Authorization & Compliance: Holding MAs, conducting pharmacovigilance
- Quality & Certification: GMP certification, pre-GMP audits
Contact us today for your individual request at service@chameleon-pharma.com!




