Pharma Production and Contract Manufacturing / GMP Consulting
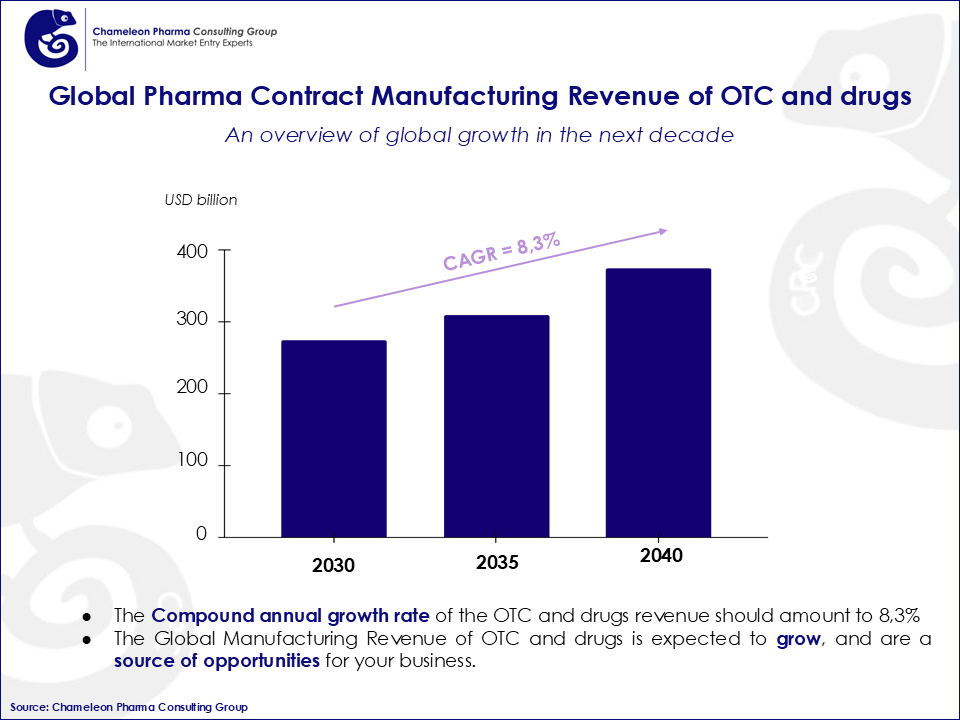
For Pharma manufacturers, staying competitive in such a strong market can be tricky. Due to high production costs and raw materials from external sources as well as changing regulatory environment, having a clear strategic vision is a must. Staying up-to-date on global pharma production trends is a necessary step to keeping your business afloat, but having deep insight into local, regional and country-specific pharma production trends can provide you with the extra push needed to the top of your market. Especially considering the impressive growth forecasts for 2040, competition in this sector is also predicted to increase so it is important to plan ahead.
Selected Production projects we at Chameleon Pharma have worked on:
- Russian GMP
- EAEU Region Regulations
- European GMP (EU-GMP)
- ANVISA GMP
- Russian/Eurasian
- GMP
- Strategic acquisition Targets
- Systematic International Partner Identification process
Advised and consulted many European and US OTC & Pharma clients on the improvements necessary to successfully completing the Russian GMP audit and receiving the RU GMP certificate.
Supported several international Consumer Health and Pharma companies to gain EAEU GMP Certificate in Russia and CIS, including the successful submission of multiple product registrations under the new EAEU Region Pharma framework.
Conducted a multi-stage analysis and developed a comprehensive roadmap to support a client and their pharmaceutical manufacturing facility in Asia in obtaining EU GMP certification.
Supported a European client in preparing for market entry into Brazil and Latin America by ensuring compliance with ANVISA GMP requirements, including a thorough audit and the successful completion of all necessary certification steps.
Advised a Medical Device client on product strategy and market alignment for the Russian and broader Eurasian region.
We have extensive experience conducting production audits to support GMP certification processes in regions including Europe, Mexico, Brazil, China, Russia, Ukraine, and others.
Identified multiple strategic acquisition targets to lower production costs through outsourcing to lower-cost Asian regions for the large-scale production of certain Consumer Health and Pharma products.
Using our Systematic International Partner Identification process, we have successfully identified international production partners to lower overall production costs.
Pharma Outsourcing Investments to Emerging Markets
More and more manufacturing contractors are building new operations in Emerging Markets like India, China, Mexico and more recently, Malaysia & Indonesia. Driven by margins, pricing, regulatory and QC pressures in developed markets, this market segment is becoming more competitive. The option to outsource certain projects to lower-cost Asian regions, especially for producing large-volume products, might be the perfect solution for Western manufacturing contractors and Pharma companies.
However, as regulations are changing and getting more difficult to pass, manufacturing processes are becoming more complex. This forces pharmaceutical players to develop longer-term and more beneficial strategies and agreements.
New ingredients and innovative packaging are key for Food supplement and cosmetic contract manufacturers
About Chameleon Pharma Consulting Group
We at Chameleon Pharma Consulting are the experts with more than 20 years of experience in areas such as Consumer Healthcare, Pharma Rx, Cosmetics, Food Supplements, FMCG, Wholesale, Retail, Production.
