Photo by David Carballar on Unsplash
Mexico is restructuring its COFEPRIS regulatory pathways to be leaner and more digital. Recent simplification measures will lead to 60% faster clinical research times and to abbreviated regulatory pathways for drugs (45 days), and medical devices (30 days). This is made possible by the progressive use of DigiPRIS, the digital platform by COFEPRIS.
What’s Changing in the COFEPRIS Process
Administrative requirements are being reduced. Effective September 1st, 2025, the Comisión Federal para la Protección contra Riesgos Sanitarios (COFEPRIS) has eliminated or merged dozens of legacy physical requirements, such as professional titles and proof of address, and consolidated multiple procedures. These changes simplify administrative handling without altering core technical dossier standards, supporting a clearer and more standardized approach to registrations, modifications, and authorizations.
With the progressive use of DigiPRIS, the digital platform from COFEPRIS, applicants can file, track, and manage parts of the lifecycle online, thus improving transparency, response coordination, and status visibility. While scope continues to evolve, the trajectory is toward broader online handling of submissions and permits.
The most impactful reform is the formal recognition of evaluations and registrations issued by international agencies such as the FDA, EMA, and PIC/S member authorities. This approach supports more predictable review timelines for eligible products and reduces duplicative regulatory steps, particularly for Rx products and medical devices.
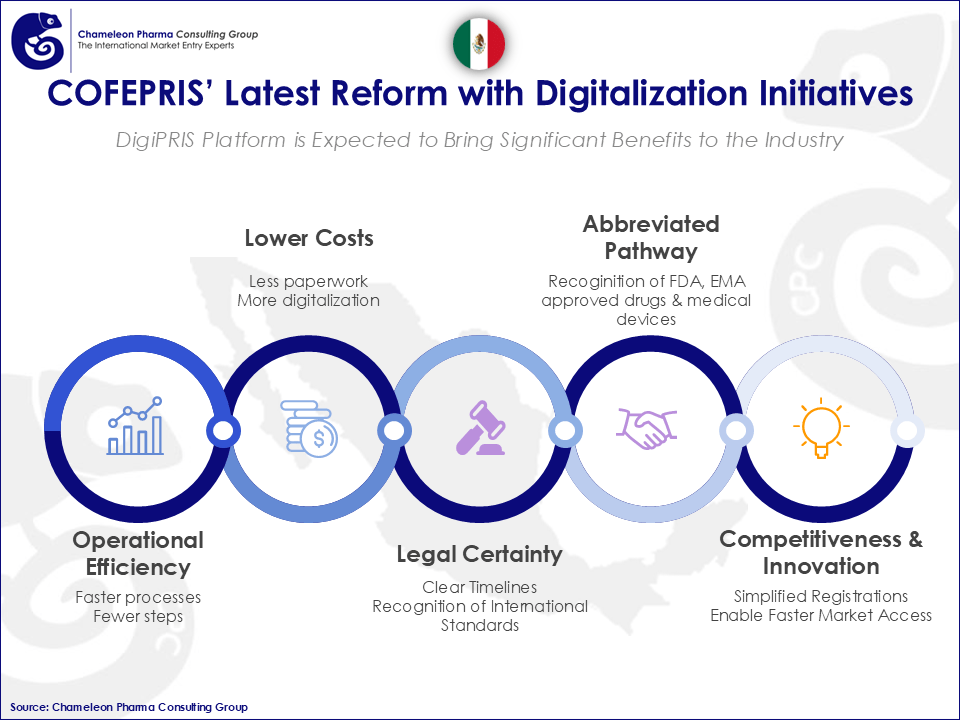
Figure 1: COFEPRIS’ Latest Reform with Digitalization Initiatives
Operational Implications for Rx, OTC, and Medical Devices
-
- Shorter, cleaner filing packages. Fewer physical annexes and merged homoclaves mean leaner document sets and simplified task lists for regulatory teams. However, regulatory teams must now ensure that digital dossiers are internally consistent and aligned with COFEPRIS’s evolving validation logic.
- More reliable tracking. Digital channels elevate transparency, enabling earlier detection of gaps and faster responses to authority queries.
- Pathway selection matters more. Where international recognitions apply, companies can leverage prior approvals to streamline Mexican submissions, which is especially relevant to Rx and higher risk devices.
- Limited retroactivity for legacy files. Applications submitted under previous rules are expected to follow the original procedural logic; most efficiency gains will accrue to new filings aligned with the updated framework.
Strategic Outlook from COFEPRIS’ Reform of Drug Registration Process
As with any major reform, implementation will vary by procedure and product type, particularly during the transition phase. Continued digitalization and process simplification are expected to strengthen Mexico’s positioning as a Latin American regulatory hub.
These changes support overall faster processes, better compliance continuity, and improved planning reliability for portfolio expansion in consumer health and Rx, as well as medical devices. Companies that adopt a digital by default submission approach and confirm eligibility for streamlined pathways early will be better placed to save time and simplify administrative work.
However, the latest reform may require some time before reaching full efficiency:
-
- Applications pending 2+ years will not benefit from the new measures, and COFEPRIS lacks a formal mechanism to withdraw already-submitted dossiers.
- Uncertainty on how COFEPRIS will handle and solve around 10,000 applications submitted before September 1st
- Minimal timeline improvement: In practice the announced timeline reductions may save only 15 business days, and the whole approval process could still take around 30 months, thus limiting the practical impact.
How Chameleon Pharma Consulting (CPC) Helps You Succeed in Mexico
Through our CPC Fast-Track Service, our regulatory experts can support your organization in navigating and operationalizing the regulatory measures in the Mexican market and help your company achieve drug registration in less than 12 months.
Moreover, we at CPC are experts in identifying local partners that fit your commercial and compliance goals. With deep regional insights, we ensure your brand enters Mexico with the right strategy, the right registrations, and the right partnerships.
Chameleon Pharma Consulting Group (CPC) has over 20 years of experience in supporting Pharma, OTC, Medical Devices, Phyto, Pharmacy compounding segment and Aesthetic Medicine companies. Having established own offices & local hubs across Latin America, Europe, Asia, the US/Canada, the Middle East, and the CEE/CIS regions is another advantage of CPC. With this local network and expertise gained from 300+ international projects and a team of 25 experts we offer our clients:
-
- Business Development, M&A, and Due Diligence
- Market Entry & Expansion: Systematic product and country analysis, market reports
- Strategic Partnering: Identifying local partners, acquisitions, or setting up own offices
- Regulatory & Registration: for drugs, MD, Derma, Aesthetic Medicine, etc.
- Market Authorization & Compliance: Holding MAs, conducting pharmacovigilance
- Quality & Certification: GMP certification, pre-GMP audits
Contact us today for your individual request at service@chameleon-pharma.com!




