Photo by Sulthan Auliya on Unsplash
Saudi Arabia stands at the forefront of healthcare transformation in the Middle East, spearheading one of the world’s most ambitious reform programs under Vision 2030. Shifting from a centralized, treatment-based model to a decentralized, preventative, and value-based system, the Kingdom has become not only the largest healthcare market in the Middle East but also one of the most dynamic and future-oriented globally.
Saudi Arabia Pharma Market Size and Growth
The Saudi pharma market has shown remarkable growth over the past decade accounting for around 32% of the entire MENA pharma market. Both patented medicines (holding a 54.4% market share) and generics (forecasted to grow to USD 5.32 billion by 2030) are driving this momentum.
On the consumer health (CH) side, demand for OTC products, vitamins, and wellness solutions is surging. This growth is fuelled by a rising middle class, expanded private insurance coverage (now reaching over 13 million people), and a societal pivot toward preventive healthcare.
The combined prescription drugs and consumer health market is expected to surpass USD 11.96 billion by 2030 and reach approximately USD 18.21 billion by 2040, provided the current regulatory and investment trends continue.
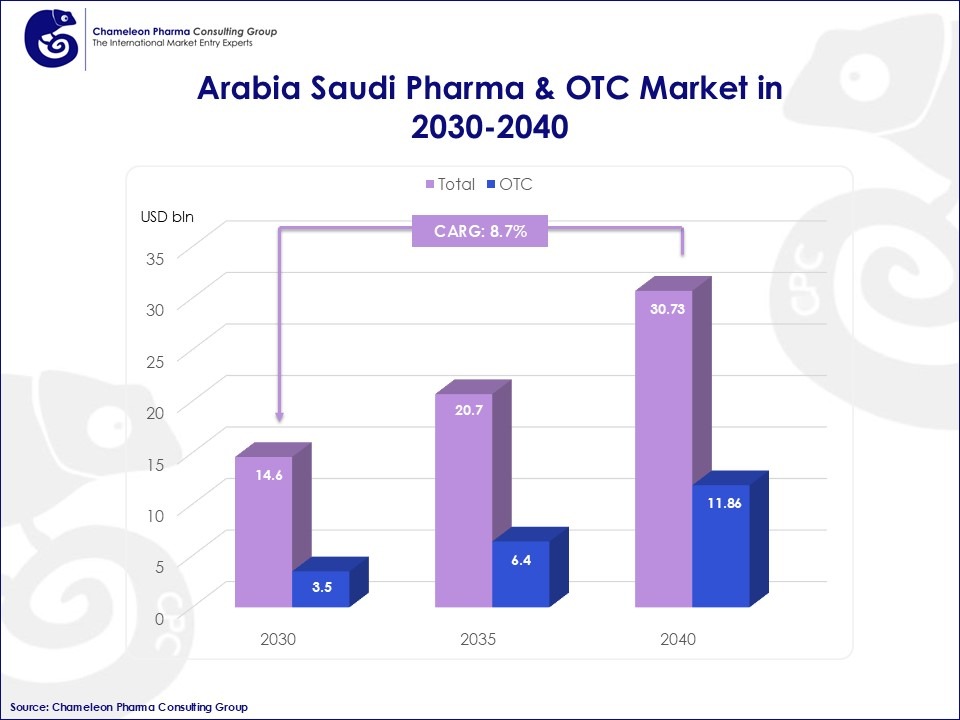
Figure 1: Arabia Saudi RX & OTC Market in 2030-40
Saudi Arabia’s Vision 2030 and Health Sector Transformation
Saudi Arabia’s Vision 2030 blueprint has brought sweeping changes to the healthcare landscape. Key milestones include:
- Establishment of the Health Holding Company and regional health clusters to decentralize healthcare delivery.
- Launch of the SEHA Virtual Hospital, now the world’s largest telemedicine platform.
- Investments of nearly USD 40 billion in infrastructure, including hospitals, clinics, supply chains, and digital health systems.
- Nationwide screening programs, early disease detection efforts, and expanded vaccination campaigns.
These initiatives are reshaping the healthcare ecosystem, emphasizing prevention, improved patient outcomes, and cost efficiency.
Regulatory Environment in Saudi Arabia
The Saudi Food and Drug Authority (SFDA) has become a global regulatory leader, achieving WHO Maturity Level 4 for medicines and vaccines regulation — a status shared only with Singapore and South Korea. This elevated regulatory maturity has positioned Saudi Arabia as a “wave-one” launch market, where companies can prioritize early product introductions alongside launches in the U.S. and Europe.
Biotech, Genomics, and Research Ambitions in Saudi Arabia
Saudi Arabia’s National Biotech Strategy sets a bold goal: to become a global leader in biopharmaceuticals and life sciences research by 2040.
- Backed by over $11 billion in planned investments, the Kingdom is channeling resources into vaccine development, gene and cell therapies, AI-driven diagnostics, and precision medicine.
- Flagship initiatives like Riyadh BioCentral and the Saudi Human Genome Project are establishing foundational infrastructure for advanced research and innovation.
- The strategy emphasizes local biomanufacturing capacity, aiming to strengthen national health security and reduce dependence on imports.
- A growing focus on personalized medicine and genomics reflects the country’s ambition to lead in next-generation healthcare technologies.
These developments align closely with Vision 2030, highlighting a government-led commitment to transforming healthcare through innovation, data, and R&D.
Investment and Private Sector Opportunities
Saudi Arabia’s evolving market has drawn major multinational players such as Roche, Pfizer, AstraZeneca, and Amgen, each expanding their local presence. Significant IPOs, including the Dr. Sulaiman Al Habib Medical Group, have injected fresh capital into the sector.
Opportunities abound for generics and branded generics manufacturers, supported by government incentives like the Value-Added Medicine guidelines and the push for local production. Additionally, niche segments — such as women’s health, fertility treatments, and chronic disease management — are poised for growth as cultural attitudes evolve and healthcare awareness deepens.
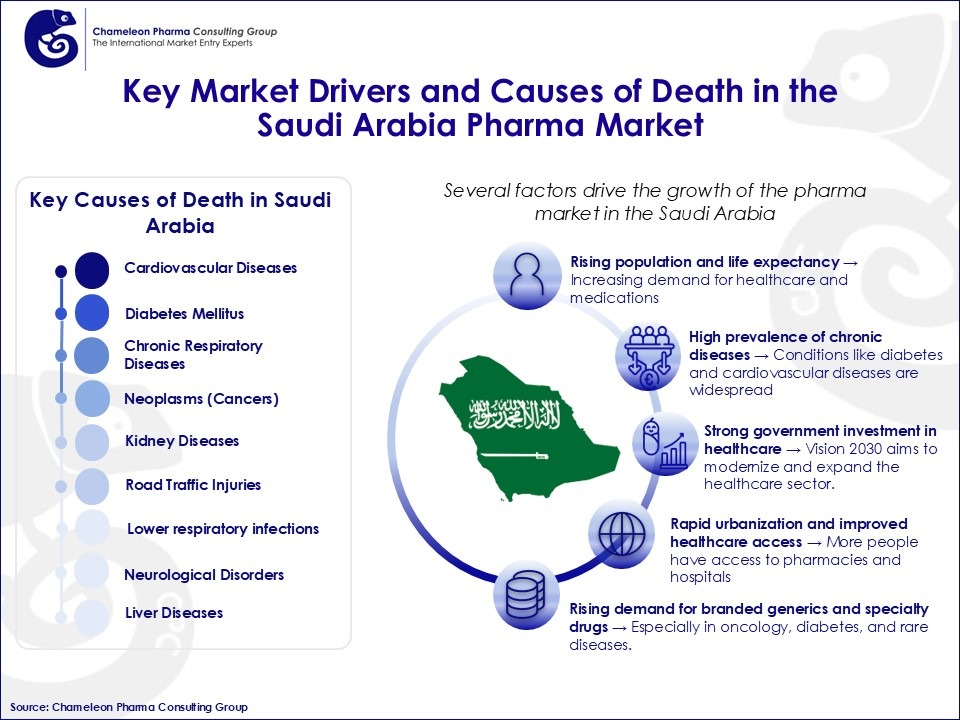
Figure 2: Key Market Drivers and Causes of Death in the Saudi Arabia Pharma Market
How CPC could assist with your MENA market entry strategy:
Chameleon Pharma Consulting Group (CPC) has over 20 years of experience in supporting Pharma, OTC, Medical Devices, Phyto, and Aesthetic Medicine companies. Having established own offices & local hubs across Latin America, Europe, Asia, the US/Canada, the Middle East, and the CEE/CIS regions is another advantage of CPC. With this local network and expertise gained from 300+ international projects and a team of 25 experts we offer our clients:
- Business Development, M&A, and Due Diligence
- Market Entry & Expansion: Systematic product and country analysis, market reports
- Strategic Partnering: Identifying local partners, acquisitions, or setting up own offices
- Regulatory & Registration: for drugs, MD, Derma, Aesthetic Medicine, etc.
- Market Authorization & Compliance: Holding MAs, conducting pharmacovigilance
- Quality & Certification: GMP certification, pre-GMP audits
Contact us today for your individual request at service@chameleon-pharma.com!




